When we think of tryptophan, the amino acid often associated with post-Thanksgiving dinner drowsiness comes to mind. But this essential nutrient plays a far more important role in your health than its turkey dinner reputation suggests—especially if you're living with chronic kidney disease (CKD).
Emerging research is showing that tryptophan metabolism is deeply tied to mood, inflammation, gut health, and immune function—and these connections are particularly important in people with CKD.
What Is Tryptophan — and What Happens When CKD Progresses?
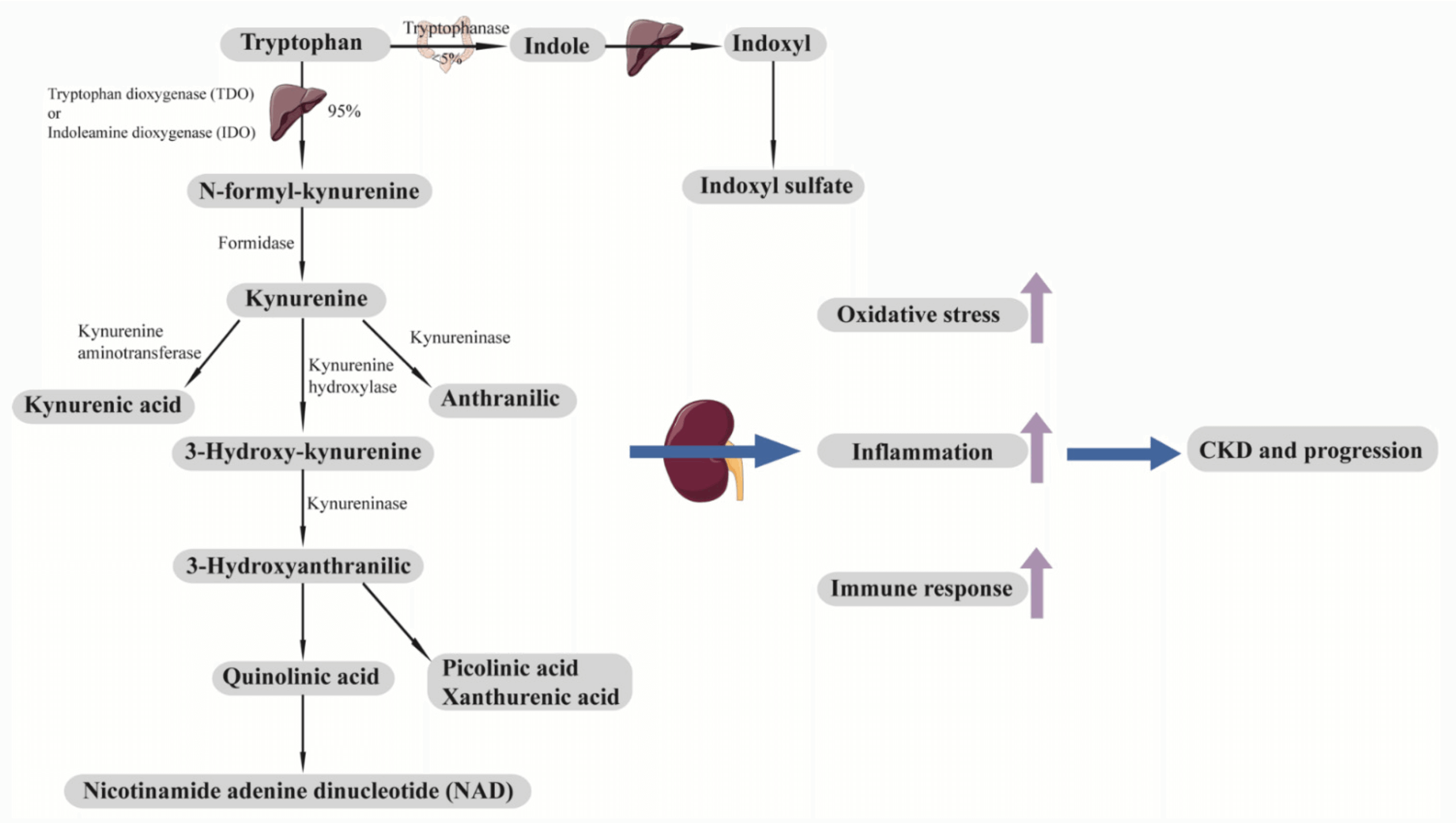
Tryptophan is an essential amino acid obtained from dietary sources such as fish, dairy, seeds, and grains. It plays a crucial role in protein synthesis and serves as a precursor for several important metabolites, including serotonin, melatonin, and various compounds produced through the kynurenine pathway [1].
As chronic kidney disease (CKD) advances, tryptophan levels in the body begin to decline. Studies have shown that in end-stage kidney disease (ESKD), tryptophan levels can drop by nearly 60% in advanced kidney disease (stage 5 CKD) compared to early stages (stage 1 CKD) [2]. This decline is gradual and correlates with the progression of CKD stages.
Why Does This Happen?
Several interconnected mechanisms contribute to declining tryptophan levels and increased metabolite accumulation in CKD:
- Reduced Elimination of Metabolites: The kidneys are responsible for clearing tryptophan and its breakdown products. As kidney function diminishes, these metabolites accumulate in the body.
Impaired Reabsorption: In healthy kidneys, amino acids like tryptophan are filtered and reabsorbed in the proximal tubules. As CKD progresses, process becomes disrupted, leading to lower circulating levels [3].
Accelerated Breakdown: Tryptophan is primarily broken down by two enzymes that become more active in CKD [1]:
IDO1 (Indoleamine 2,3-dioxygenase 1) – activated by inflammatory cytokines such as IL-6 and TNF-α. In CKD, persistent inflammation stimulates IDO1 activity, accelerating tryptophan degradation through the kynurenine pathway.
TDO (Tryptophan 2,3-dioxygenase) – mainly regulated by hormonal and dietary factors in the liver.
Chronic Inflammation: A hallmark of CKD, systemic inflammation contributes not only to cardiovascular and immune dysfunction but also accelerates tryptophan depletion by upregulating IDO1 activity.
This shift in tryptophan metabolism, especially through inflammation-driven enzyme activity, leads us directly to the kynurenine pathway — the main route by which tryptophan is broken down in the body.
The Three Pathways of Tryptophan Metabolism
Once absorbed from food, tryptophan can be metabolized in several different directions — but not all of them are beneficial, especially in people with CKD.
Only about 1% of dietary tryptophan is used for building proteins. The remaining 99% is funneled into five major metabolic pathways. In the context of CKD, three of these are especially important [4]:
1. The Kynurenine Pathway (≈90% of tryptophan)
This is the dominant route of tryptophan breakdown in the body via enzymes like indoleamine dioxygenase (IDO) and tryptophan dioxygenase (TDO). It produces a range of biologically active compounds such as:
- Kynurenine
- Quinolinic acid
- Kynurenic acid
- Nicotinamide (NAD)
In CKD, inflammation upregulates enzymes like IDO1, accelerating this pathway and increasing the production of these metabolites.
2. The Serotonin Pathway (≈1–2%)
A small portion of tryptophan is converted into serotonin and melatonin, which influence mood, sleep, and gut motility.
3. The Indole Pathway (≈4–6%)
This pathway occurs in the gut and is driven by gut microbes. In the colon, bacterial enzymes convert tryptophan into indoles, which are then absorbed and transformed in the liver into various metabolites.
Some indole derivatives — such as indole-3-propionic acid (IPA) and indole-3-aldehyde — are beneficial and help support gut barrier integrity, reduce inflammation, and modulate immune function.
Others — like indole-3-acetic acid (IAA) and the uremic toxin indoxyl sulfate (IS) — can become harmful when overproduced, especially in the setting of gut dysbiosis and reduced kidney clearance [5].

What Are the Consequences?
As tryptophan levels fall in CKD, its breakdown products — including kynurenine, kynurenic acid, and quinolinic acid — accumulate due to impaired kidney clearance. These metabolites contribute to a range of complications that can affect how patients feel and function.
Fatigue & Depression: Elevated levels of kynurenine and its derivatives are associated with neurological effects like fatigue, depression, and cognitive impairment in CKD patients [1].
Gut Health Impact: Disruptions in gut microbiota can shift tryptophan metabolism toward harmful metabolites like indoxyl sulfate, which impair intestinal barrier function and drive inflammation [5].
Immune Dysregulation: In CKD, elevated tryptophan catabolites like indoxyl sulfate may disrupt immune cell signaling via the aryl hydrocarbon receptor (AhR) and pregnane X receptor (PXR), contributing to immune dysfunction and inflammation [5].
Increased inflammation: Kynurenine pathway metabolites — including kynurenine, quinolinic acid, and 3-hydroxykynurenine — promote oxidative stress and inflammation, contributing to CKD progression [1].
Higher Cardiovascular Risks: Higher concentrations of kynurenine have been linked to increased cardiovascular events and mortality [6]. An elevated kynurenine-to-tryptophan ratio (KTR) is also associated with a higher risk of heart failure in CKD [7].
If you're dealing with any of these symptoms, nutrition might be playing a bigger role than you realize. Talk to a CKD dietitian about how to support your tryptophan metabolism.
Image from: https://www.mdpi.com/2073-4409/10/11/2832
Depression and CKD: More Than Just the Diagnosis
Depression is the most common mental health condition in people with CKD, affecting up to 25% overall and as many as 73% in end-stage kidney disease (ESKD)—far higher than the 5% seen in the general adult population [4].
These high rates aren't just due to the emotional toll of living with a chronic illness. Biological changes in CKD play a key role, particularly disruptions in tryptophan metabolism.
As kidney function declines, toxic tryptophan byproducts like kynurenines and indoxyl sulfate accumulate in the blood. These compounds lower serotonin levels in the brain by up to 40%, drive inflammation, and interfere with brain signaling through receptors like AhR. Together, these effects may explain why depression is so prevalent—and often more challenging to address—in people with CKD [4].
Potential Treatment Approaches
Researchers are exploring several strategies to address tryptophan metabolism in CKD, including:
Targeting the Kynurenine Pathway: Therapies targeting IDO1 and TDO enzymes to regulate tryptophan degradation are under investigation [1].
Biotic interventions: Prebiotics, probiotics, and synbiotics to restore microbial balance [5].
Dietary fiber: To promote short-chain fatty acids (SCFAs), which support the gut lining and reduce systemic inflammation [5].
Individualized protein intake: Lowering excessive protein in later CKD stages may reduce harmful metabolites without creating amino acid deficiencies [2]. However, CKD is also associated with tryptophan depletion, so a personalized approach should include strategic inclusion of tryptophan-rich foods within your recommended protein limits.
Nutritional Strategies: What You Can Do
Supporting tryptophan metabolism in CKD isn’t about a quick fix. It’s about strategic, whole-person nutrition that supports the gut, reduces inflammation, and maintains amino acid balance. Consider:
Moderate protein intake: Not too much, not too little.
Include tryptophan-rich foods:
Pumpkin seeds (~110 mg per ¼ cup)
Oatmeal (~75 mg per 1/2 cup cooked)
Tofu (~72 mg per 1 oz)
Edamame (~53 mg per ¼ cup cooked)
Milk (~100 mg per 1 cup)
Turkey or Chicken (~80-95 mg per oz)
Fish (e.g. salmon, cod, ~65-90 mg per oz)
Egg (~78 mg each)
More fiber: Make it a habit to regularly include vegetables, fruits, whole grains, nuts and seeds, as well as supplementation (as recommended) with prebiotics like FOS, inulin, and acacia gum.
Not sure where to start with probiotics, fiber, or adjusting your protein? Get expert support from our KNI dietitians trained in advanced CKD care.

Final Thoughts
Tryptophan metabolism is one of those under-the-radar topics that’s becoming increasingly important in CKD care. As kidney function declines, tryptophan levels drop while harmful byproducts like kynurenine and indoxyl sulfate build up — and this can contribute to some of the most frustrating symptoms CKD patients face: fatigue, mood changes, inflammation, and even cardiovascular and gut issues. These changes stem from chronic inflammation, impaired clearance of toxins, shifts in gut microbiota, and disruptions in how the body processes protein and amino acids.
The good news is that this is an area where nutrition and gut health strategies can make a meaningful difference. Supporting your tryptophan metabolism through the right protein intake, more dietary fiber, and even probiotics or prebiotics may help reduce toxic metabolite buildup and support better overall well-being. Understanding this pathway doesn’t just add to the science — it opens new, practical options for feeling better with CKD.
If you’re curious about how this applies to your personal situation, working with a renal dietitian can help you navigate it safely and effectively. You can connect with a CKD expert at the Kidney Nutrition Institute to take the next step in building a personalized nutrition plan that supports your kidney health.
**Note: Always consult with a healthcare provider or dietitian before making significant dietary changes, especially in the context of CKD.
References
Mor A, Kalaska B, Pawlak D. Kynurenine Pathway in Chronic Kidney Disease: What's Old, What's New, and What's Next?. Int J Tryptophan Res. 2020;13:1178646920954882. Published 2020 Sep 21. doi:10.1177/1178646920954882
Paats J, Adoberg A, Arund J, Dhondt A, Fernström A, Fridolin I, Glorieux G, Leis L, Luman M, Gonzalez-Parra E, et al. Serum Levels and Removal by Haemodialysis and Haemodiafiltration of Tryptophan-Derived Uremic Toxins in ESKD Patients. International Journal of Molecular Sciences. 2020; 21(4):1522. https://doi.org/10.3390/ijms21041522
Farrell P. Renal Transporters: Biochemistry, Physiology, Pharmacology, Pathology. The Medical Biochemistry Page. Updated March 2024. Accessed April 22, 2025. https://themedicalbiochemistrypage.org/renal-transporters-biochemistry-physiology-pharmacology-pathology/
Lefrère A, Burtey S, Bobot S, Belzeaux R, Bobot M. Depression in chronic kidney disease: Particularities, specific mechanisms and therapeutic considerations, a narrative review. Behav Brain Res. 2025;483:115467. https://doi.org/10.1016/j.bbr.2025.115467
Madella AM, Van Bergenhenegouwen J, Garssen J, Masereeuw R, Overbeek SA. Microbial-Derived Tryptophan Catabolites, Kidney Disease and Gut Inflammation. Toxins. 2022;14(9):645. https://doi.org/10.3390/toxins14090645
El Chamieh C, Larabi IA, Alencar De Pinho N, et al. Study of the association between serum levels of kynurenine and cardiovascular outcomes and overall mortality in chronic kidney disease. Clin Kidney J. 2023;17(1):sfad248. Published 2023 Sep 28. doi:10.1093/ckj/sfad248
- Mohiti S, Boachie C, Minović I, et al. Serum tryptophan and kynurenine levels and risk of heart failure among patients with chronic kidney disease. Clin Nutr. 2025;47:14–20. doi:10.1016/j.clnu.2025.01.028

Work With Us
Our proprietary Kidney Health Method is the framework that we use with our patients to see HUGE results! We can do the same for you. To start feeling better NOW, book an appointment with one of our renal dietitians!